P3 E) States of Matter
There are two changes that may happen to the particles in a substance when a substance is heated or cooled. These changes are:
- The substance will heat up or cool down, which will change the amount of energy that the particles have in their kinetic energy stores. If a substance is heated up, the particles will have more energy in their kinetic energy stores and will therefore move around faster. If a substance cools down, the particles will have less energy in their kinetic energy stores and therefore move around slower.
- The energy change (either increase or decrease) may change the intermolecular bonds that hold the particles in a substance together. This changes the potential energy stores. The potential energy stores for a particular substance is the greatest in a gas, then a liquid and the least in a solid.
When we are heating a substance within a state, the heat energy is transferred to the kinetic energy stores of the particles, which increases the total internal energy and temperature of the substance. Specific heat capacity explores how increasing the amount of energy in a substance increases the internal temperature of the substance within a state (click here for more information on specific heat capacity).
When a substance is at its melting or boiling point and we continue to add heat energy to the substance, the energy is used to break the intermolecular bonds that are holding the particles together, which results in a change in state. This increases the total internal energy of a substance, but it does not increase the temperature of the substance. Specific latent heat explores how increasing the amount of energy in a substance that is at its melting or boiling point breaks the intermolecular bonds that are holding the particles together resulting in a change in state (click here for more information on specific latent heat).
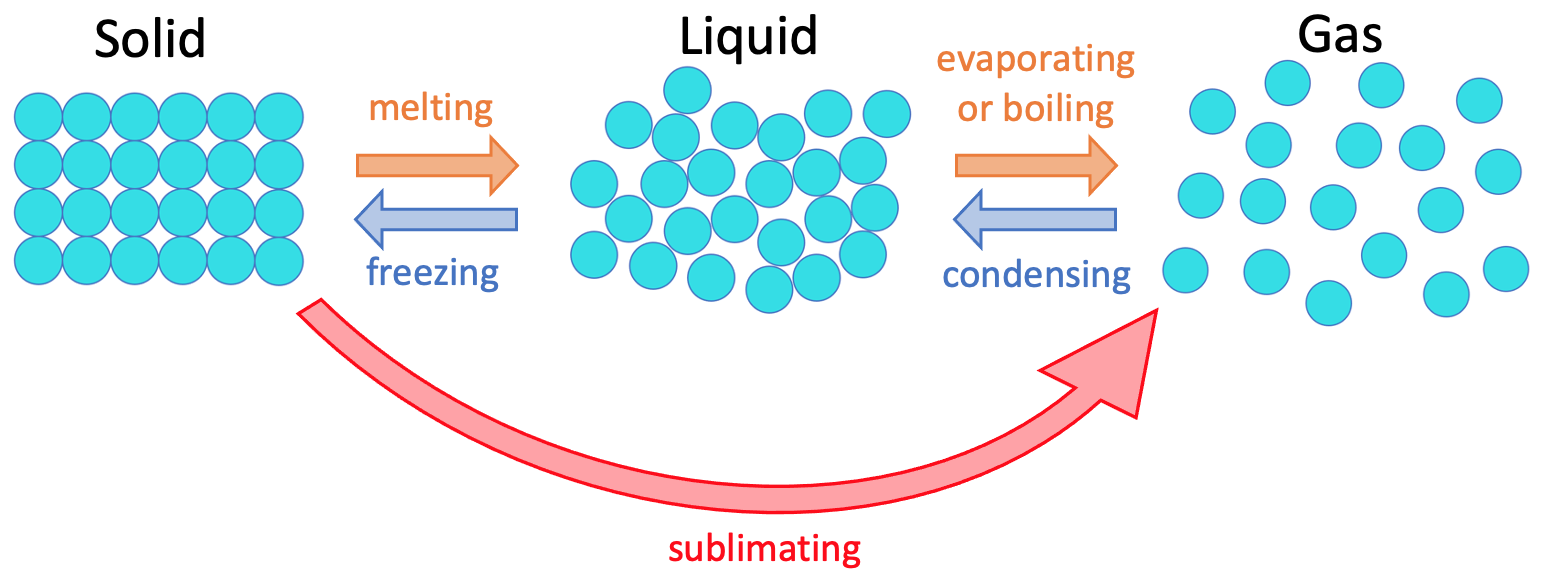
There are 3 different states of matter; solid, liquid and gas. We can change a solid to a liquid and then a gas by heating up the substance. We can change a gas to a liquid and then a solid by cooling a substance down. The different states and the names for the changes between the different states are shown below.
When a substance changes state, the substance is exactly the same substance, just in a different state. This means that a change in state is a physical change and not a chemical change (a chemical change would be when we end up with a difference substance).
The number of particles in a substance is exactly the same when a state changes. If we had an ice cube (solid water; H2O) and heated it up which resulted in it melting to become liquid water, and then boiling to become water vapour (gas), we would have the same number and type of particles (all H2O) in all of the 3 different states. This means that the mass of the substance is the same in the 3 different states.
The difference between the 3 states is the arrangement of the particles and the amount of energy that the particles have in their internal energy stores (kinetic energy stores and potential energy stores).