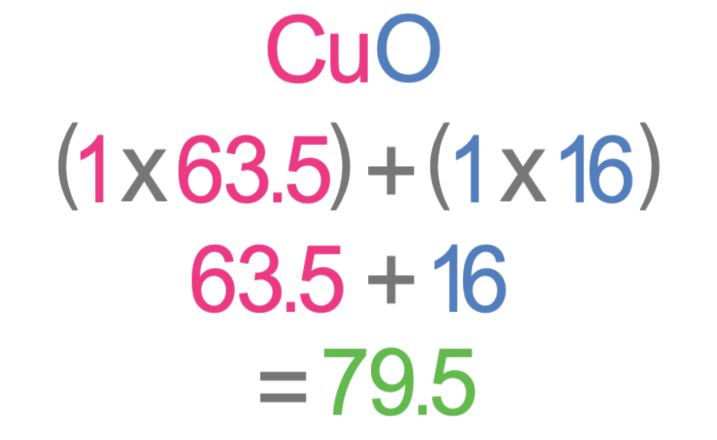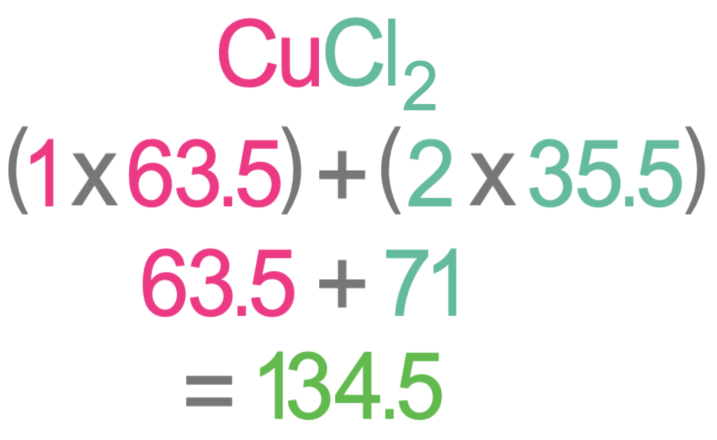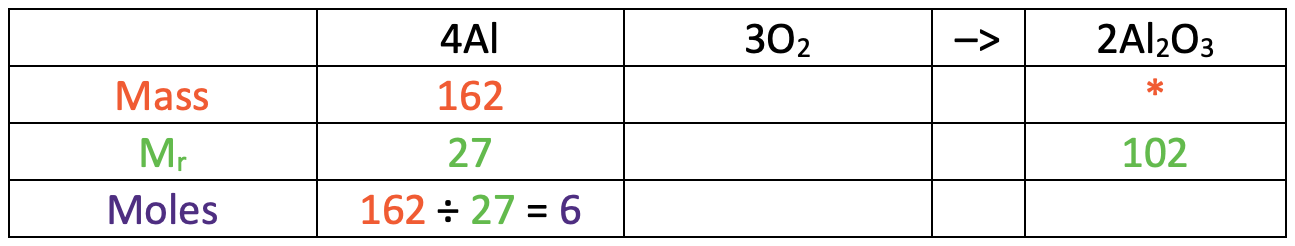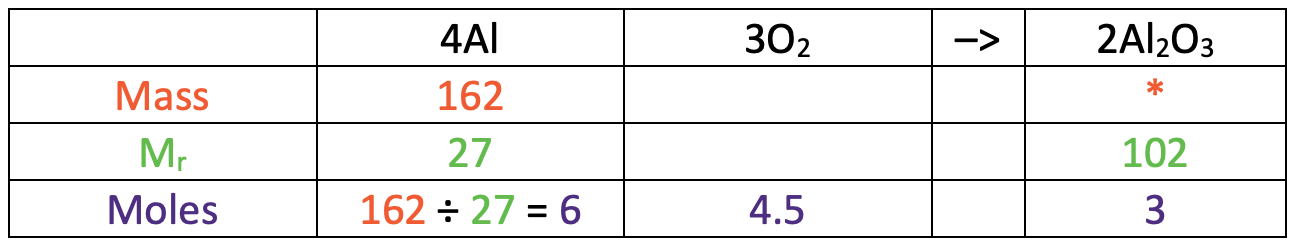C3 F) Moles & Equations
Reactions stop when one of the reactants is used up – the reactant that is used up is known as the limiting reactant because it limits/ stops reactions taking place when it is used up. All of the other reactants in the reaction will be known as being in excess. The reactants that are in excess are usually added in excess on purpose to ensure that all of the limiting reactant fully reacts.
For example, let’s suppose that we are burning 144 g of magnesium in air (oxygen) to give us magnesium oxide. The balanced symbol equation is shown below:
The amount of the products formed will be in direct proportion to the amount of the limiting reactant used. If we double the amount of the limiting reactants (in this case double the amount of magnesium), we would get double the quantity of the products (magnesium oxide) providing that magnesium is still the limiting factor (which it will be because there is plenty of oxygen in air). Alternatively, if we halved the amount of the limiting reactant, we would get half of the products.
For the above reaction, we can see that 2 moles of magnesium will react with 1 mole of oxygen to produce 2 moles of magnesium oxide. The ratio from left to right would be 2 : 1 : 2.
- If we were to double the amount of magnesium, we would double the ratio; the ratio from left to right would be 4 : 2 : 4. This would mean that 4 moles of magnesium will react with 2 moles of oxygen to produce 4 moles of magnesium oxide.
- If we were to half the original amount of magnesium and only had 1 mole of magnesium, we would halve the original ratio (2 : 1 : 2) to get the ratio 1 : 0.5 : 1. This would mean that 1 mole of magnesium will react with 0.5 moles of oxygen to produce 1 mole of magnesium oxide
In this section we are going to have a look at a few questions whereby we are asked to calculate the mass of a product formed from a given mass of a limiting reactant. We use the moles formula triangle when answering these questions and this formula triangle is shown below.
144 grams of magnesium is burnt in air (oxygen) to give us magnesium oxide.
The balanced symbol equation is shown below.
The Ar of magnesium is 24 and the Ar of oxygen is 16.
Calculate the mass of magnesium oxide produced.
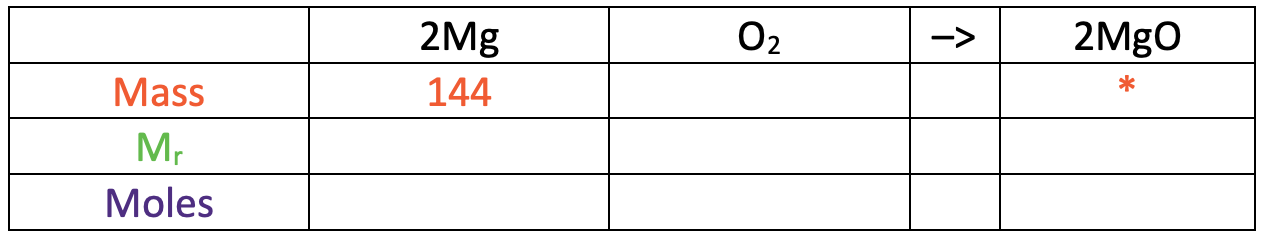
I am now going to turn the reaction equation into a table where each of the rows will be mass, relative formula mass (Mr) and moles.
In the table above, I have also added some information that we are given/ want to find. We were told in the question that the mass of magnesium was 144 g and we want to find what the mass of magnesium oxide is (I have added a star in this cell).
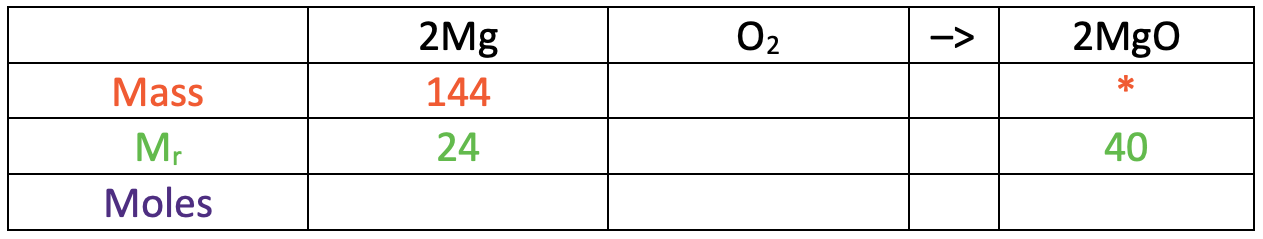
We now need to work out the relative formula mass (Mr) or relative atomic mass (Ar) of both magnesium and magnesium oxide.
The relative atom mass of magnesium is 24.
Magnesium oxide is made out of 1 magnesium atom and 1 oxygen atom. The Ar of magnesium is 24 and the Ar of oxygen is 16. We work out the Mr of magnesium oxide by adding 1 magnesium and 1 oxygen together. The working is shown below.
The Mr of magnesium oxide is 40.
I have added the Ar for magnesium (24) and the Mr for magnesium oxide (40) to the table.
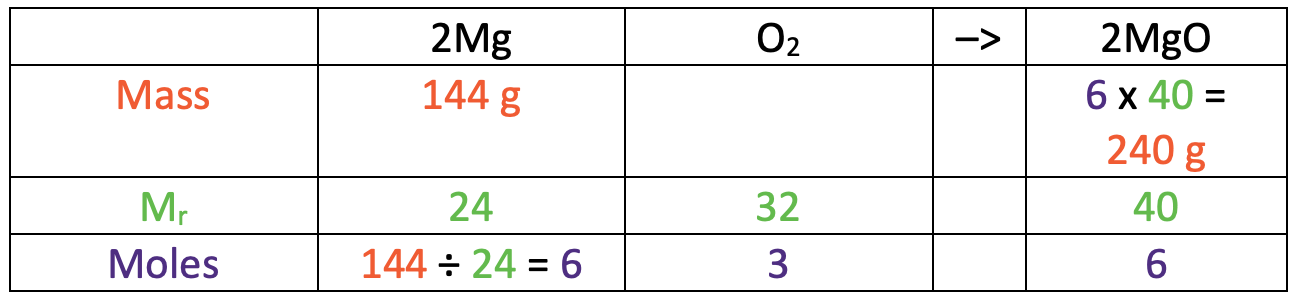
The next step is to find the number of moles of magnesium that we have. We work out the number of moles of a substance by dividing the mass (144 g) by the relative formula/ atomic mass (24). The working is shown in the table below.
This tells us that we have 6 moles of magnesium. We now use the ratio from the balanced symbol equation to find the number of moles of magnesium oxide that we have; we use the ratio from the big numbers in front of the molecules in the balanced symbol equation. For this reaction, we can see that 2 moles of magnesium will react with 1 mole of oxygen to produce 2 moles of magnesium oxide. The ratio from left to right is 2 : 1 : 2.
We found that there are 6 moles of magnesium, which is 3 times the amount in the ratio. Therefore, our ratio from left to right is 6 : 3 : 6. I have added these values to the table.
We now have the number of moles for magnesium oxide (6) and the relative formula mass for magnesium oxide (40). This means that we can work out the mass of magnesium oxide by multiplying the number of moles (6) by the relative formula mass (40). The working is shown in the table below.
This tells us that the mass of magnesium oxide that we would get is 240 g.
We can extend this example to show that the law of conservation of mass holds. The law of conservation of mass states that the mass of all of the reactants will be the same as the mass of all of the products. This is because no atoms are created or destroyed during the reaction.
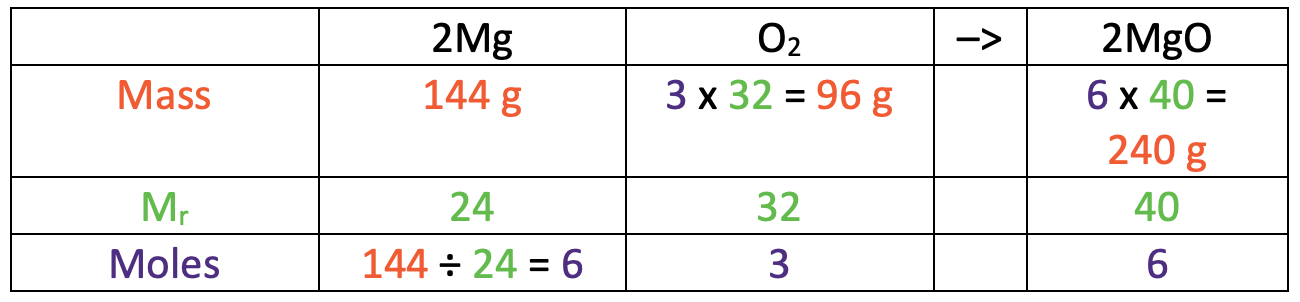
In order to prove that the law of conservation of mass holds, we need to find the mass of oxygen (O2). Before we can find the mass of oxygen, we need to find the Mr of oxygen (O2). Oxygen (O2) is made out of 2 oxygen atoms, and each of these oxygen atoms has an Ar of 16. Therefore, we work out the Mr of oxygen (O2) by multiplying 2 (the number of oxygen atoms in O2) by 16 (the Ar of oxygen), which gives us 32 (2 x 16 = 32). I have added an Mr of 32 for oxygen to the table below.
We work out the mass of oxygen by multiplying the number of moles (3) by the relative formula mass (32). The working is shown in the table below.
We can now work out the mass of the reactants, and the mass of the products, and check that they are equal to each other.
- The mass of reactants is the mass of magnesium (144 g) and the mass of oxygen (96 g) added together, which gives us 240 g (144 + 96 = 240).
- This reaction only has one product, which is magnesium oxide and it has a mass of 240 g
From these calculations, we can see that the mass of the reactants and products are the same; they are both 240 g. This shows us that the law of conservation of mass holds.
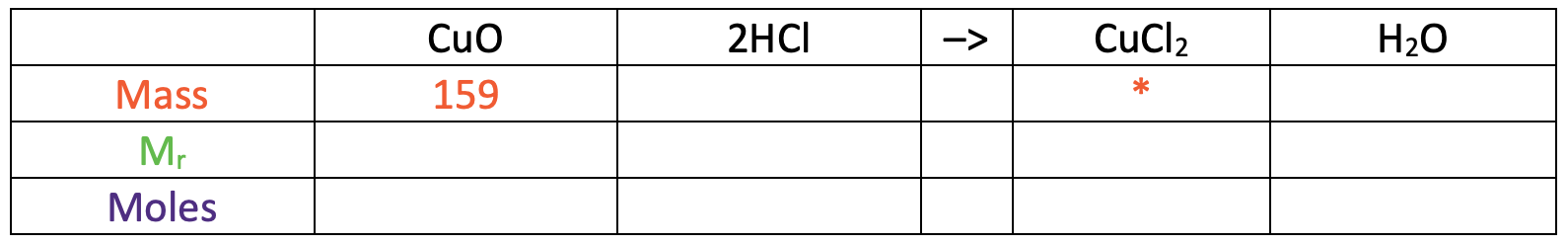
We are reacting 159 g of copper oxide with excess hydrochloric acid. The balanced equation for this reaction is shown below.
Copper has an Ar of 63.5, oxygen has an Ar of 16, hydrogen has an Ar of 1 and chorine has an Ar of 35.5.
Calculate the mass of copper chloride produced.
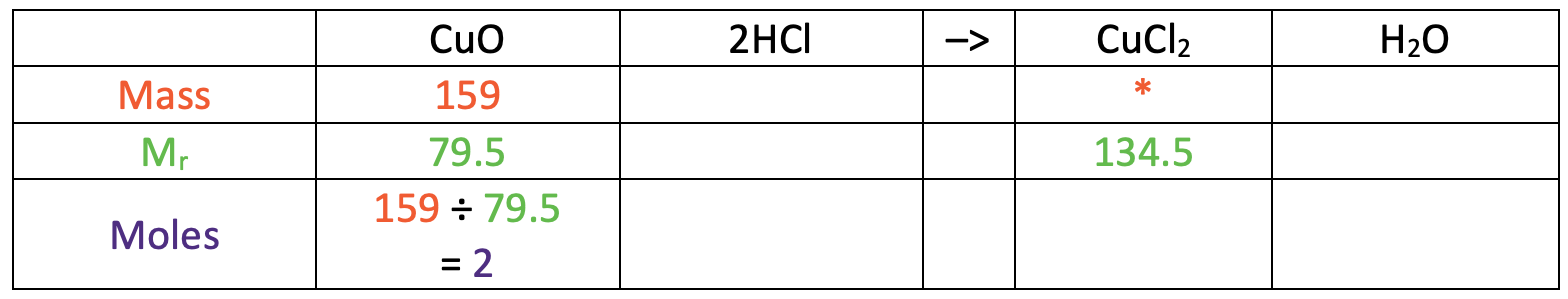
I am now going to turn the reaction equation into a table where each of the rows will be mass, relative formula mass (Mr) and moles.
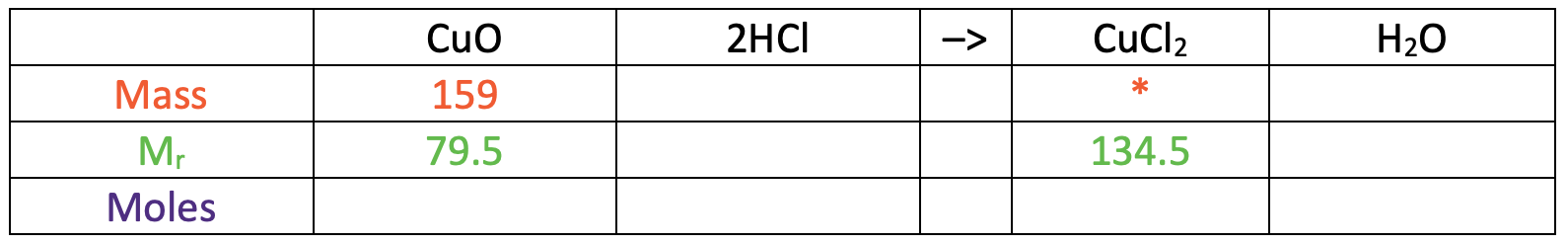
In the table above, I have also added some information that we are given/ want to find. We were told in the question that the mass of copper oxide used was 159 g and we want to find the mass of copper chloride (I have added a star in this cell).
We now need to work out the relative formula mass (Mr) of copper oxide and copper chloride. In order to work out the relative formula mass of copper oxide and copper chloride, we need to know the relative atomic mass of each of the elements. We were told in the question that copper has an Ar of 63.5, oxygen has an Ar of 16 and chorine has an Ar of 35.5. The working for the relative formula mass of copper oxide and copper chloride is shown below.
The relative formula mass for copper oxide is 79.5 and the relative formula mass for copper chloride is 134.5. I have added these values to the table.
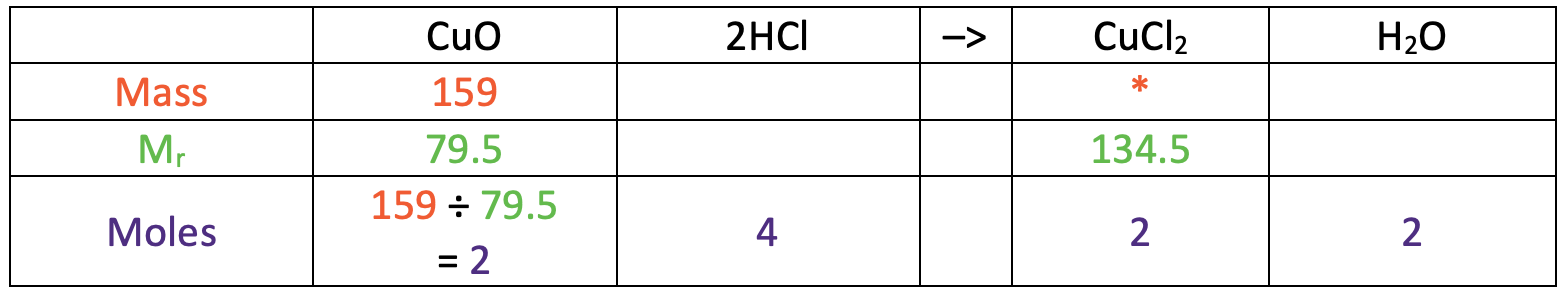
The next step is to find the number of moles of copper oxide that we have. We work out the number of moles of a substance by dividing the mass (159 g) by the relative formula mass (79.5). The working is shown in the table below.
This tells us that we have 2 moles of copper oxide.
We now use ratios to work out the number of moles of copper chloride that we have. From the balanced equation, we can see that 1 mole of copper oxide will react with 2 moles of hydrochloric acid to produce 1 mole of copper chloride and 1 mole of water; the ratio of the substances from left to right is 1 : 2 : 1 : 1. We have 2 moles of copper oxide, which is twice the amount in the ratio. This means that we double our substances in the ratio to get the ratio from left to right as 2 : 4 : 2 : 2. I have added these values to the table.
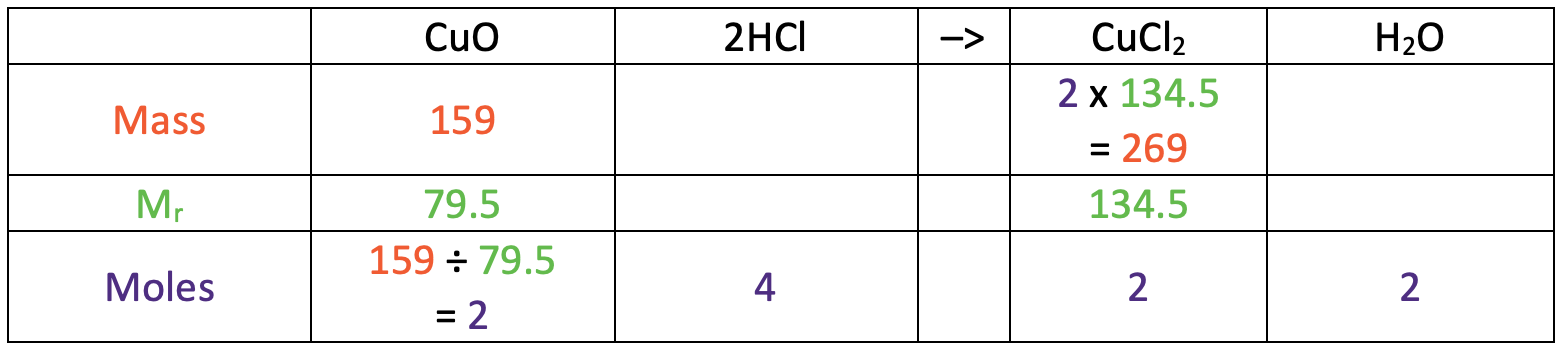
We now have the number of moles for copper chloride (2) and the relative formula mass for copper chloride (134.5). This means that we can work out the mass for copper chloride by multiplying the number of moles (2) by the relative formula mass (134.5). The working is shown in the table below.
This tells us that the mass of copper chloride is 269 g.
162 grams of aluminium is burnt in air (oxygen) to give aluminium oxide.
The balanced symbol equation is shown below.
The Ar of aluminium is 27 and the Ar of oxygen is 16.
Calculate the mass of magnesium oxide produced.
I am now going to turn the reaction equation into a table where each of the rows will be mass, relative formula mass (Mr) and moles.
In the table above, I have also added some information that we are given/ want to find. We were told in the question that the mass of aluminium was 162 g and we want to find out what the mass of aluminium oxide is (I have added a star in this cell).
We now need to work out the relative formula mass (Mr) or relative atomic mass (Ar) of both aluminium and aluminium oxide.
The relative atomic mass of aluminium is 24.
Aluminium oxide (Al2O3) is made out of 2 aluminium atoms and 3 oxygen atoms. The Ar of aluminium is 27 and the Ar of oxygen is 16. We work out the Mr of aluminium oxide by adding 2 aluminiums and 3 oxygens together. The working for this is shown below.
This tells us that the relative formula mass of aluminium oxide is 102.
I have added the Ar for aluminium (27) and the Mr for aluminium oxide (102) to the table below.
The next step is to find the number of moles of aluminium that we have. We work out the number of moles of a substance by dividing the mass (162 g) by the relative formula mass (27). The working is shown in the table below.
This tells us that we have 6 moles of aluminium.
We now use the ratio from the balanced symbol equation to find the number of moles of aluminium oxide that we have; we use the ratio from the big numbers in front of the molecules. For this reaction, we can see that 4 moles of aluminium will react with 3 moles of oxygen to produce 2 moles of aluminium oxide. The ratio from left to right is 4 : 3 : 2.
We found that there are 6 moles of aluminium, which is 1.5 times the amount in the ratio. Therefore, we multiply our ratio by 1.5 to get the ratio 6 : 4.5 : 3. I have added these values to the table.
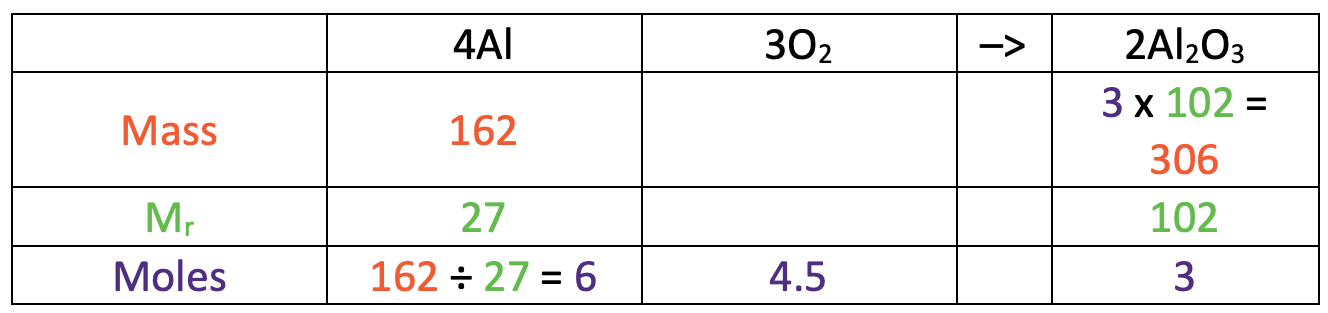
We now have the number of moles of aluminium oxide (3) and the relative formula mass for aluminium oxide (102). This means that we can work out the mass of aluminium oxide by multiplying the number of moles (3) by the relative formula mass (102). The working is shown in the table below.
This tells us that the mass of aluminium oxide that we would get is 306 g.
The content in this section is quite tricky. Therefore, you may need to work through the examples a few times before you feel comfortable in answering these types of questions.