Back to C1 Home
C1: Quiz 2 – Answers
C1: Quiz 2 – Answers
1)
a) Relative atomic mass
b) Atomic number or proton number or atomic (proton) number
c) P = 11, N = 12, E = 11
d)
a) Relative atomic mass
b) Atomic number or proton number or atomic (proton) number
c) P = 11, N = 12, E = 11
d)
2)
a) P = 13, N = 14, E = 13
b)
3)
a) P = 20, N = 20, E = 20
b)
4) The groups tell us the number of electrons in the outermost electron shell of an atom. The periods tell us the number of electron shells that the atom has.
5)
a) 5 electrons in its outermost shell
b) 2 electron shells
c)
6)
7)
Similarity – they both have 2 electron shells as they are both in period 2
Difference – they have a different number of electrons in their outermost shells; boron has 3 electrons in its outermost shell as it is in group 3, and fluorine has 7 electrons in its outermost shell as it is in group 7.
8)
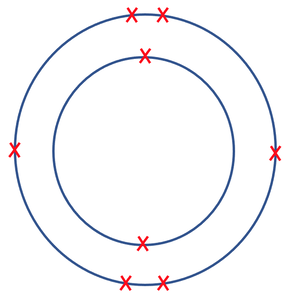
a) The atom is in group 5 because it has 5 electrons in its outermost shell
b) The atom is in period 3 as it has 3 electron shells
c) Phosphorus
Explanation – we are told that the element is neutrally charged, which means that it has the same number of protons as electrons; there are 15 electrons on the atom, which means that the atom will have 15 protons. Phosphorus has 15 protons (or an atomic number of 15).
9)
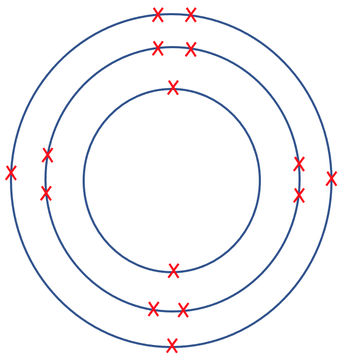
a) The atom is in group 6 because it has 6 electrons in its outermost shell
b) The atom is in period 2 as it has 2 shells
c) Oxygen
Explanation – we are told that the element is neutrally charged, which means that it has the same number of protons as electrons; there are 8 electrons on the atom, which means that the atom will have 8 protons. Oxygen has 8 protons (or an atomic number of 8).
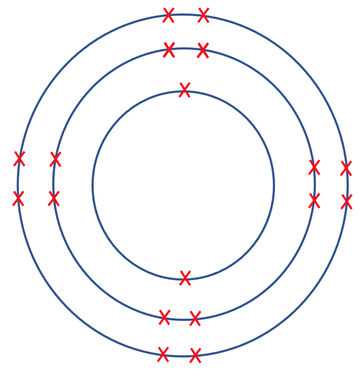
10) Sulfur
Explanation – the atom has 18 electrons. The question tells us that it has a charge of 2-, and this means that the atom has 2 more electrons than protons. Therefore, as the atom has 18 electrons, it must have 16 protons. According to the periodic table, the atom with 16 protons is sulfur
Similarity – they both have 2 electron shells as they are both in period 2
Difference – they have a different number of electrons in their outermost shells; boron has 3 electrons in its outermost shell as it is in group 3, and fluorine has 7 electrons in its outermost shell as it is in group 7.
8)
a) The atom is in group 5 because it has 5 electrons in its outermost shell
b) The atom is in period 3 as it has 3 electron shells
c) Phosphorus
Explanation – we are told that the element is neutrally charged, which means that it has the same number of protons as electrons; there are 15 electrons on the atom, which means that the atom will have 15 protons. Phosphorus has 15 protons (or an atomic number of 15).
9)
a) The atom is in group 6 because it has 6 electrons in its outermost shell
b) The atom is in period 2 as it has 2 shells
c) Oxygen
Explanation – we are told that the element is neutrally charged, which means that it has the same number of protons as electrons; there are 8 electrons on the atom, which means that the atom will have 8 protons. Oxygen has 8 protons (or an atomic number of 8).
10) Sulfur
Explanation – the atom has 18 electrons. The question tells us that it has a charge of 2-, and this means that the atom has 2 more electrons than protons. Therefore, as the atom has 18 electrons, it must have 16 protons. According to the periodic table, the atom with 16 protons is sulfur
Questions
Click here for a printable version of this quiz.
1) The tile below is for sodium.
Click here for a printable version of this quiz.
1) The tile below is for sodium.
a) What is the name of the top number (the 23)?
b) What is the name of the bottom number (the 11)?
c) How many protons, neutrons and electrons does sodium have?
d) Draw the electronic structure of a sodium atom.
b) What is the name of the bottom number (the 11)?
c) How many protons, neutrons and electrons does sodium have?
d) Draw the electronic structure of a sodium atom.
2) The tile below is for aluminium.
a) How many protons, neutrons and electrons does aluminium have?
b) Draw the electronic structure of an aluminium atom.
b) Draw the electronic structure of an aluminium atom.
3) The tile below is for calcium.
a) How many protons, neutrons and electrons does calcium have?
b) Draw the electronic structure of a calcium atom.
b) Draw the electronic structure of a calcium atom.
4) What is the difference between the groups and the periods in the periodic table?
5) Nitrogen is in group 5 and period 2.
a) How many electrons will be in nitrogen’s outermost shell?
b) How many electron shells will nitrogen have?
c) Draw the electronic structure of nitrogen.
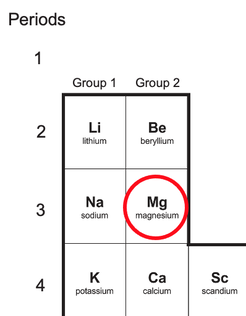
6) The image below shows you part of a periodic table without the relative atomic masses and atomic numbers.
Draw the electronic structure of a magnesium atom.
7) Boron is in period 2 and group 3.
Fluorine is in period 2 and group 7.
Give one similarity and one difference between the electronic structures of boron and fluorine. Explain your answers.
8) The diagram below shows the electronic structure for a neutral atom.
7) Boron is in period 2 and group 3.
Fluorine is in period 2 and group 7.
Give one similarity and one difference between the electronic structures of boron and fluorine. Explain your answers.
8) The diagram below shows the electronic structure for a neutral atom.
a) What group is this atom in and why?
b) What period is this atom in and why?
c) Use a periodic table to find out which atom this is. Click here to see a periodic table.
9) The diagram below shows the electronic structure for a neutral atom.
b) What period is this atom in and why?
c) Use a periodic table to find out which atom this is. Click here to see a periodic table.
9) The diagram below shows the electronic structure for a neutral atom.
a) What group is this atom in and why?
b) What period is this atom in and why?
c) Use a periodic table to find out which atom this is.
10) The atom below has a charge of 2-.
b) What period is this atom in and why?
c) Use a periodic table to find out which atom this is.
10) The atom below has a charge of 2-.
Use a periodic table to find out which atom this is.