Paper 3 H - SAMPLE SET 1 Q7
7) Ammonium nitrate (NH4NO3) is produced by reacting ammonia with nitric acid.
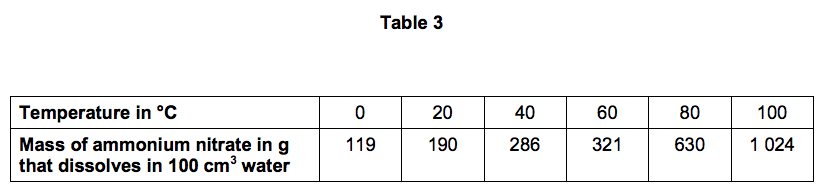
A student measured the mass of ammonium nitrate that dissolves in 100 cm3 of water at different temperatures.
Table 3 shows the student’s results.
A student measured the mass of ammonium nitrate that dissolves in 100 cm3 of water at different temperatures.
Table 3 shows the student’s results.
7.1) Use Table 3 to plot a graph of the solubility of ammonium nitrate on Figure 9.
[4 marks]
7.2) At 20 °C, 190 g of ammonium nitrate dissolves in 100 cm3 of water.
Calculate the amount of ammonium nitrate (in moles) that dissolves in 1 dm3 of water at 20 °C.
Relative atomic masses (Ar): H = 1; N = 14; O = 16 [3 marks]
Calculate the amount of ammonium nitrate (in moles) that dissolves in 1 dm3 of water at 20 °C.
Relative atomic masses (Ar): H = 1; N = 14; O = 16 [3 marks]
Amount of dissolved ammonium nitrate = _______________ mol
7.3) Farmers use ammonium nitrate as a fertiliser.
Farmers want to slow down the rate at which ammonium nitrate fertiliser dissolves in the water in the soil.
Suggest why they spread the fertiliser in the form of small beads instead of a fine powder.
[2 marks]
7.4) Ammonia is needed to make ammonium nitrate.
The reaction used to make ammonia is:
The forward reaction is exothermic.
At equilibrium, about 35% of the nitrogen and hydrogen are converted to ammonia at 450 °C and 200 atmospheres pressure.
Explain the effects of increasing the temperature, or increasing the pressure, on the amount of ammonia produced at equilibrium. [4 marks]
(Total for Question 7 = 13 marks)
At equilibrium, about 35% of the nitrogen and hydrogen are converted to ammonia at 450 °C and 200 atmospheres pressure.
Explain the effects of increasing the temperature, or increasing the pressure, on the amount of ammonia produced at equilibrium. [4 marks]
(Total for Question 7 = 13 marks)