Paper 3 H - SAMPLE SET 1 Q6
6) Aluminium is produced from an ore called bauxite.
Bauxite contains aluminium oxide.
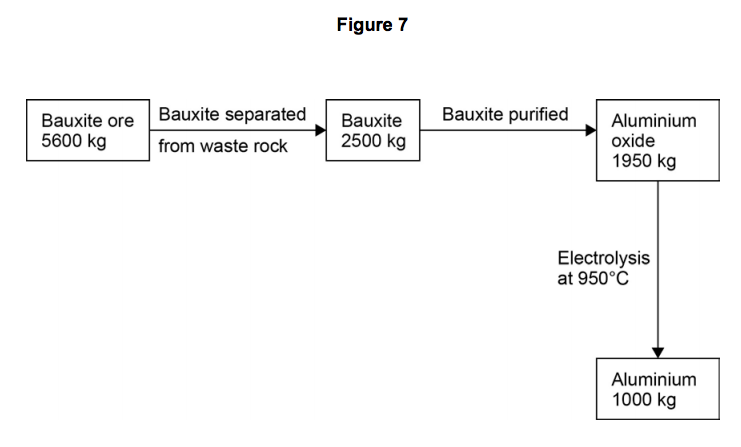
Look at Figure 7.
Bauxite contains aluminium oxide.
Look at Figure 7.
6.1) Calculate the percentage of bauxite that is converted into aluminium oxide.
[2 marks]
Percentage = __________________
6.2) Show by calculation that the mass of aluminium produced is less than that expected from 1 950 kg aluminium oxide (Al2O3).
You should state the difference in the mass of aluminium expected and the mass of aluminium produced to three significant figures.
Relative atomic masses (Ar): O = 16; Al = 27 [3 marks]
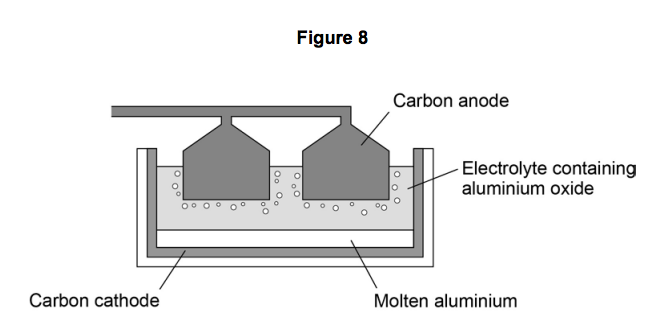
Figure 8 shows an electrolysis cell used to extract aluminium.
6.3) Why does the carbon anode used in the electrolysis cell need to be continually replaced?
[3 marks]
6.4) In an electrolysis cell the current is 1.5 × 105 A, at a potential difference of 4V.
Calculate the energy transferred by the electrolysis cell in 24 hours. [5 marks]
6.4) In an electrolysis cell the current is 1.5 × 105 A, at a potential difference of 4V.
Calculate the energy transferred by the electrolysis cell in 24 hours. [5 marks]
Energy transferred = _________________ J
6.5) The half equation at the cathode is:
Calculate the number of moles of electrons needed to produce 1 000 kg of aluminium.
Give your answer to three significant figures.
Relative atomic mass (Ar): Al = 27 [3 marks]
Give your answer to three significant figures.
Relative atomic mass (Ar): Al = 27 [3 marks]
Answer = ________________ moles
(Total for Question 6 = 16 marks)