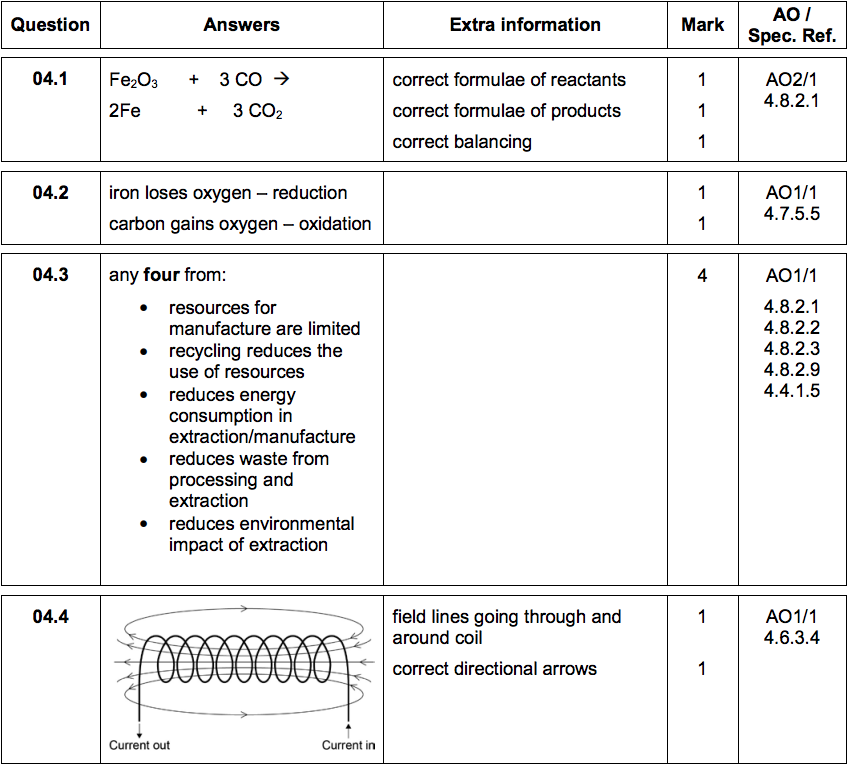
Paper 3 H - SAMPLE SET 1 Q4 Answers
4) Iron is a metal that has many uses.
4.1) Iron is extracted from iron ore. Part of the process involves reduction of the ore with carbon monoxide. Iron ore contains iron oxide (Fe2O3).
Write a balanced equation for the reaction of iron oxide with carbon monoxide. [3 marks]
4.2) Explain why this reaction is a redox reaction. [2 marks]
Steel is an alloy of iron. Steel is used to make cars.
After its useful life a car is taken to a scrapyard for recycling.
4.3) Suggest four benefits of recycling a car body. [4 marks]
Figure 5 shows an electromagnet being used to lift a car in a scrapyard.
4.1) Iron is extracted from iron ore. Part of the process involves reduction of the ore with carbon monoxide. Iron ore contains iron oxide (Fe2O3).
Write a balanced equation for the reaction of iron oxide with carbon monoxide. [3 marks]
4.2) Explain why this reaction is a redox reaction. [2 marks]
Steel is an alloy of iron. Steel is used to make cars.
After its useful life a car is taken to a scrapyard for recycling.
4.3) Suggest four benefits of recycling a car body. [4 marks]
Figure 5 shows an electromagnet being used to lift a car in a scrapyard.
4.4) An electromagnet is made up of a solenoid.
Figure 6 shows a solenoid.
Figure 6 shows a solenoid.
Draw the magnetic field of the solenoid on Figure 6. [2 marks]
4.5) In a scrapyard, an electromagnet is used to lift and release cars so they can be moved around.
Suggest two ways a solenoid could be made to lift and release cars in a scrapyard.
Explain why each suggestion would be useful in the scrapyard. [4 marks]
(Total for Question 4 = 15 marks)
4.5) In a scrapyard, an electromagnet is used to lift and release cars so they can be moved around.
Suggest two ways a solenoid could be made to lift and release cars in a scrapyard.
Explain why each suggestion would be useful in the scrapyard. [4 marks]
(Total for Question 4 = 15 marks)